PRODUCT IDENTIFICATION
H.S. CODE
TOXICITY
CLASSIFICATION
PHYSICAL AND CHEMICAL PROPERTIES
AUTOIGNITION
NFPA RATINGS
REFRACTIVE INDEX
APPLICATIONS
FATTY ACID CONTENT
99.0% min
GENERAL DESCRIPTION OF LONG CHAIN UNSATURATED FATTY ACID
|
COMMON NAME |
SYSTEMATIC NAME |
CAS RN |
SYMBOL |
| Myristolenic Acid |
(Z)-9-Tetradecenoic Acid |
544-64-9 |
14:1 n-5 |
| Palmitoleic Acid | (Z)-9-Hexadecenoic Acid | 373-49-9 |
16:1 n-7 |
| Palmitelaidic Acid | (E)-9-Hexadecenoic Acid | 10030-73-6 |
16;1 n-7 (trans) |
| Vaccenic Acid | (Z)-11-Octadecenoic Acid | 506-17-2 | 18:1 n-7 |
|
Oleic Acid |
(Z)-9-Octadecenoic Acid |
112-80-1 | 18:1 n-9 |
| Elaidic Acid | (E)-9-Octadecenoic Acid | 112-79-8 | 18:1 n-9 (trans) |
| Linoelaidicic Acid | (E,E)-9,12-Octadecadienoic Acid | 506-21-8 |
18:2 n-6 (trans) |
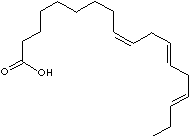
| Linoleic Acid | (Z,Z)-9,12-Octadecadienoic Acid | 60-33-3 |
18:2 n-6 |
| Isolinoleic Acid | (E,E)-9,11-Octadecadienoic Acid | 544-71-8 |
18:2 n-6 (trans) |
| Linolenic Acid | (Z,Z,Z)-9,12,15-Octadecatrienoic Acid | 463-40-1 | 18:3 n-3 |
| gamma -Linolenic Acid | (Z,Z,Z)-6,9,12-Octadecatrienoic Acid | 506-26-3 | 18:3 n-6 |
|
Moroctic Acid |
(Z,Z,Z,Z)-6,9,12,15-Octadecatetraenoic Acid |
20290-75-9 |
18:4 n-3 |
| Eicosenoic Acid |
(Z)-11-Ecosenoic Acid |
5561-99-9 | 20:1 n-9 |
| Eicosadienoic Acid |
(Z,Z,)-11,14-Ecosadienoic Acid |
2091-39-6 | 20:2 n-6 |
| Mead Acid |
(Z,Z,Z)-5,8,11-Eicosatrienoic Acid |
20590-32-3 | 20:3 n-9 |
|
Eicosatrienoic Acid |
(Z,Z,Z)-11,14,17-Eicosatrienoic Acid |
17046-59-2 |
20:3 n-3 |
| Dihomo-g -Linolenic Acid |
(Z,Z,Z)-8,11,14-Eicosatrienoic Acid |
1783-84-2 | 20:3 n-6 |
| Omega-3 Arachidonic Acid |
(Z,Z,Z,Z)-8,11,14,17-Eicosatetraenoic Acid |
24880-40-8 | 20:4 n-3 |
| Arachidonic Acid |
(Z,Z,Z,Z)-5,8,11,14-Eicosatetraenoic Acid |
506-32-1 | 20:4 n-6 |
| Timnodonic Acid | (Z,Z,Z,Z,Z)-5,8,11,14,17-Eicosapentaenoic Acid | 10417-94-4 | 20:5 n-3 |
| Erucic Acid |
(Z)-13-Docosenoic Acid |
112-86-7 |
22:1 n-9 |
| Docosadienoic Acid |
(Z,Z)-13,16-Docosadienoic Acid |
17735-98-7 |
22:2 n-6 |
| Docosatrienoic Acid |
(Z,Z,Z)-13,16,19-Docosatrienoic Acid |
28845-86-5 |
22:3 n-3 |
| Adrenic Acid |
(Z,Z,Z,Z)-7,10-13-16-Ocosatetraenoic Acid |
28874-58-0 |
22:4 n-6 |
| Docosapentaenoic Acid |
(Z,Z,Z,Z,Z)-4,7,10,13,16-Docosapentaenoic Acid |
25182-74-5 |
22:5 n-6 |
| Docosapentaenoic Acid |
(Z,Z,Z,Z,Z)-7,10,13,16,19-Docosapentaenoic Acid |
24880-45-3 |
22:5 n-3 |
| Docosahexaenoic Acid |
(Z,Z,Z,Z,Z,Z)-4,7,10,13,16,19-Docosahexaenoic Acid |
6217-54-5 |
22:6 n-3 |
| Nervonic Acid |
(Z)-15-Tetracosaenoic Acid |
506-37-6 | 24:1 n-9 |
| Tetracosahexaenoic Acid |
((Z,Z,Z,Z,Z,Z)-6,9,12,15,18,21-Tetracosahexaenoic Acid |
24:6 n-3 |